-
摘要:目的
探讨BRCA1/2突变乳腺癌FOXP3表达情况及潜在意义。
方法选取北京大学肿瘤医院48例BRCA突变者(BRCA1 16例,BRCA2 32例)和78例年龄匹配的非突变者,采用免疫组织化学检测乳腺癌组织FOXP3的表达情况。为验证免疫组织化学结果,分析TCGA-BRCA中39例BRCA1、36例BRCA2和948例非携带者乳腺癌的FOXP3 RNA表达水平及其与同源重组缺陷评分的关系。
结果在BRCA1突变者中FOXP3阳性率43.8%(7/16),BRCA2突变者为59.4%(19/32),非携带者为9.0%(7/78)。BRCA1/2突变患者FOXP3阳性率均显著高于非突变者(P=0.002;P<0.001)。TCGA-BRCA结果显示,BRCA1/2突变乳腺癌的FOXP3 RNA水平也显著高于非携带者(P=0.02,P=0.004)。FOXP3 RNA水平与同源重组缺陷评分正相关(Spearman R=0.30, P<2.2e-16)。
结论BRCA1/2突变乳腺癌较非突变者FOXP3表达更高,可能对免疫治疗更敏感。
Abstract:ObjectiveTo investigate the potential significance of FOXP3 expression in BRCA1/2-mutant breast cancer.
MethodsA total of 48 BRCA mutation carriers (16 with BRCA1 and 32 with BRCA2) and 78 age-matched non-carriers were included in this study. Immunohistochemistry was used to detect the expression of FOXP3 in breast cancer tissues. The FOXP3 RNA expression in 39 BRCA1, 36 BRCA2, and 948 non-carrier breast cancer patients from TCGA-BRCA and the correlation with homologous recombination deficiency scores were evaluated to validate the immunohistochemistry results.
ResultsThe FOXP3 positive rate was 43.8% (7/16) in BRCA1 mutation carriers, 59.4% (19/32) in BRCA2 mutation carriers, and 9.0% (7/78) in non-carriers. The FOXP3 positive rates in patients with BRCA1/2 mutant breast cancer were significantly higher than those in non-carriers (P=0.002; P<0.001). TCGA-BRCA results showed that the FOXP3 RNA level in BRCA1/2 mutant breast cancer was significantly higher than that in non-carriers (P=0.02, P=0.004). The FOXP3 RNA level was positively correlated with the homologous recombination deficiency score (Spearman R=0.30, P<2.2e-16).
ConclusionPatients with BRCA1/2 mutant breast cancers have higher FOXP3 expression than non-carriers, and may be more sensitive to immunotherapy.
-
Key words:
- BRCA1/2 mutation /
- Breast cancer /
- FOXP3 /
- Homologous recombination deficiency
-
0 引言
乳腺癌是全球最常见的女性恶性肿瘤[1],其中BRCA突变乳腺癌约占5%[2-3]。BRCA1/2胚系突变乳腺癌发病年龄早、双侧乳腺癌发生率较高、家族聚集现象明显,BRCA1突变乳腺癌中三阴性乳腺癌(triple negative breast cancer, TNBC)占比高达60%[4]。近年来,免疫治疗逐渐成为乳腺癌新的治疗策略,尤其是针对三阴性乳腺癌,抗PD-1/PD-L1单克隆抗体已经在临床试验中显示出一定的疗效[5-6]。BRCA1/2突变乳腺癌具有较高的肿瘤突变负荷,以及较强的T淋巴细胞浸润和免疫检查点分子PD-1/PD-L1的表达[7-9],BRCA1/2突变乳腺癌是否也是免疫治疗的获益人群值得研究。
FOXP3(forkhead box P3)是一种转录因子,其基因位于X染色体的短臂上,位点为Xp.11.23[10-11]。FOXP3主要表达于调节性T细胞(regulatory T cells, Tregs),是Tregs的特异性标志物和功能决定因子,FOXP3的表达水平可以反映Tregs的数量和功能[10-11]。Tregs是一类具有免疫抑制功能的CD4+ T细胞,通过产生抑制性细胞因子(如白介素10)或以细胞间接触依赖性方式调节T细胞或抗原呈递细胞,从而抑制肿瘤特异性的免疫反应,促进肿瘤的免疫逃逸,降低免疫治疗的效果[12-13]。因此,FOXP3作为Tregs的关键分子,被认为是一种重要的免疫治疗相关的生物标志物。
但是目前BRCA1/2突变乳腺癌中FOXP3的表达情况并不清楚。本研究通过临床样本进行免疫组织化学检测,并利用TCGA-BRCA数据进一步验证,以明确BRCA1/2突变携带者和非携带者乳腺癌中FOXP3的表达情况,以及FOXP3表达水平与基因组不稳定性的关系。
1 资料与方法
1.1 研究对象
本研究纳入2003年1月至2022年7月北京大学肿瘤医院收治的48例BRCA胚系突变乳腺癌患者(BRCA1 16例,BRCA2 32例)和78例年龄匹配的非突变携带者。患者外周血样本DNA用于检测BRCA1/2基因的突变状态,患者手术后乳腺癌组织样本进行石蜡包埋。所有的患者均为女性,详细的临床信息见表1。通过北京大学肿瘤医院电子病历系统及随访办公室获得患者的病史信息,肿瘤大小为术前B型超声检查提示的肿瘤最大径。淋巴结状态根据前哨淋巴结活检和(或)腋窝淋巴结清扫病理结果进行判断。雌激素受体(estrogen receptor, ER)、孕激素受体(progesterone receptor, PR)和人表皮生长因子受体2(human epidermal growth factor receptor 2, HER2)的状态根据术前穿刺或手术样本免疫组织化学染色结果确定。ER和PR阳性定义为≥1%的肿瘤细胞染色阳性;HER2阳性定义为HER2免疫组织化学染色3+,或HER2免疫组织化学染色2+,同时荧光原位杂交(fluorescence in situ hybridization, FISH)实验显示HER2基因扩增。ER或PR阳性统称为激素受体(hormone receptor, HR)阳性。本研究得到了北京大学肿瘤医院研究与伦理委员会的批准,均签署患者知情同意书。
表 1 BRCA1/2突变乳腺癌患者和非携带者临床病理特征Table 1 Clinicopathological characteristics of patients with BRCA1/2-mutant breast cancer and non-carriersCharacteristics No. BRCA1 carriers (n(%)) BRCA2 carriers (n(%)) Non-carriers (n(%)) P1 P2 P3 Total 126 16 32 78 Median follow-up
time (range, years)4.7 (0.1-19.2) 8.2 (1.5-19.2) 9.2 (0.1-18.2) 4.0 (0.4-5.1) Age at diagnosis (years) Median (range) 49 (27-75) 43.5 (28-64) 49.5 (31-74) 50 (27-75) ≤ 50 68 12 (75.0%) 16 (50.0%) 40 (51.3%) 0.14 >0.999 0.18 > 50 58 4 (25.0%) 16 (50.0%) 38 (48.7%) ER status Positive 74 5 (31.3%) 24 (75.0%) 45 (57.7%) 0.10 0.14 0.009 Negative 52 11 (68.8%) 8 (25.0%) 33 (42.3%) PR status Positive 65 5 (31.3%) 26 (81.3%) 34 (43.6%) 0.52 < 0.001 0.002 Negative 61 11 (68.8%) 6 (18.8%) 44 (56.4%) HER2 status Positive 26 1 (6.3%) 3 (9.4%) 22 (28.2%) 0.11 0.06 >0.999 Negative 100 15 (93.8%) 29 (90.6%) 56 (71.8%) Molecular type TNBC 35 11 (68.8%) 4 (12.5%) 20 (25.6%) 0.01 0.008 < 0.001 HR+ and HER2− 65 4 (25.0%) 25 (78.1%) 36 (46.2%) HER2+ 26 1 (6.3%) 3 (9.4%) 22 (28.2%) Tumor stage Ⅰ 29 5 (31.3%) 9 (28.1%) 15 (19.2%) 0.21 0.23 0.67 Ⅱ 77 9 (56.3%) 14 (43.8%) 54 (69.2%) Ⅲ 12 0 (0) 3 (9.4%) 9 (11.5%) Unknown 8 2 (12.5%) 6 (18.8%) 0 (0) Tumor size (cm) ≤2 50 6 (37.5%) 19 (59.4%) 25 (32.1%) 0.56 0.01 0.35 >2 75 9 (56.3%) 13 (40.6%) 53 (67.9%) Unknown 1 1 (6.3%) 0 (0) 0 (0) Nodal status Positive 41 4 (25.0%) 12 (37.5%) 25 (32.1%) 0.77 0.79 0.59 Negative 85 12 (75.0%) 20 (62.5%) 53 (67.9%) Notes: P1: BRCA1 carriers versus non-carriers; P2: BRCA2 carriers versus non-carriers; P3: BRCA1 carriers versus BRCA2 carriers. ER: estrogen receptor; PR: progesterone receptor; HER2: human epidermal growth factor receptor 2; TNBC: triple-negative breast cancer; HR: hormone receptor. 1.2 免疫组织化学检测FOXP3表达情况
所有乳腺癌组织在4%中性福尔马林固定后石蜡包埋。免疫组织化学实验采用EnVision法。具体步骤如下:60℃烤片1 h,经过二甲苯脱蜡、梯度酒精水合后,用磷酸盐缓冲液(phosphate buffered saline, PBS)洗涤。3%双氧水封闭10 min,EDTA高压修复10 min,冷却后用5%羊血清封闭1 h,一抗孵育过夜。第二天PBS洗涤后,二抗孵育0.5 h,再次洗涤后进行DAB显色,苏木精对比染色1 min,盐酸-酒精分色液分色数秒,流水返蓝15 min,最后进行梯度酒精脱水、中性树胶封片。一抗为1:500 FOXP3(ab20034,小鼠单克隆抗体,Abcam)。二抗为酶标羊抗鼠/兔IgG聚合物(基因科技上海股份有限公司)。
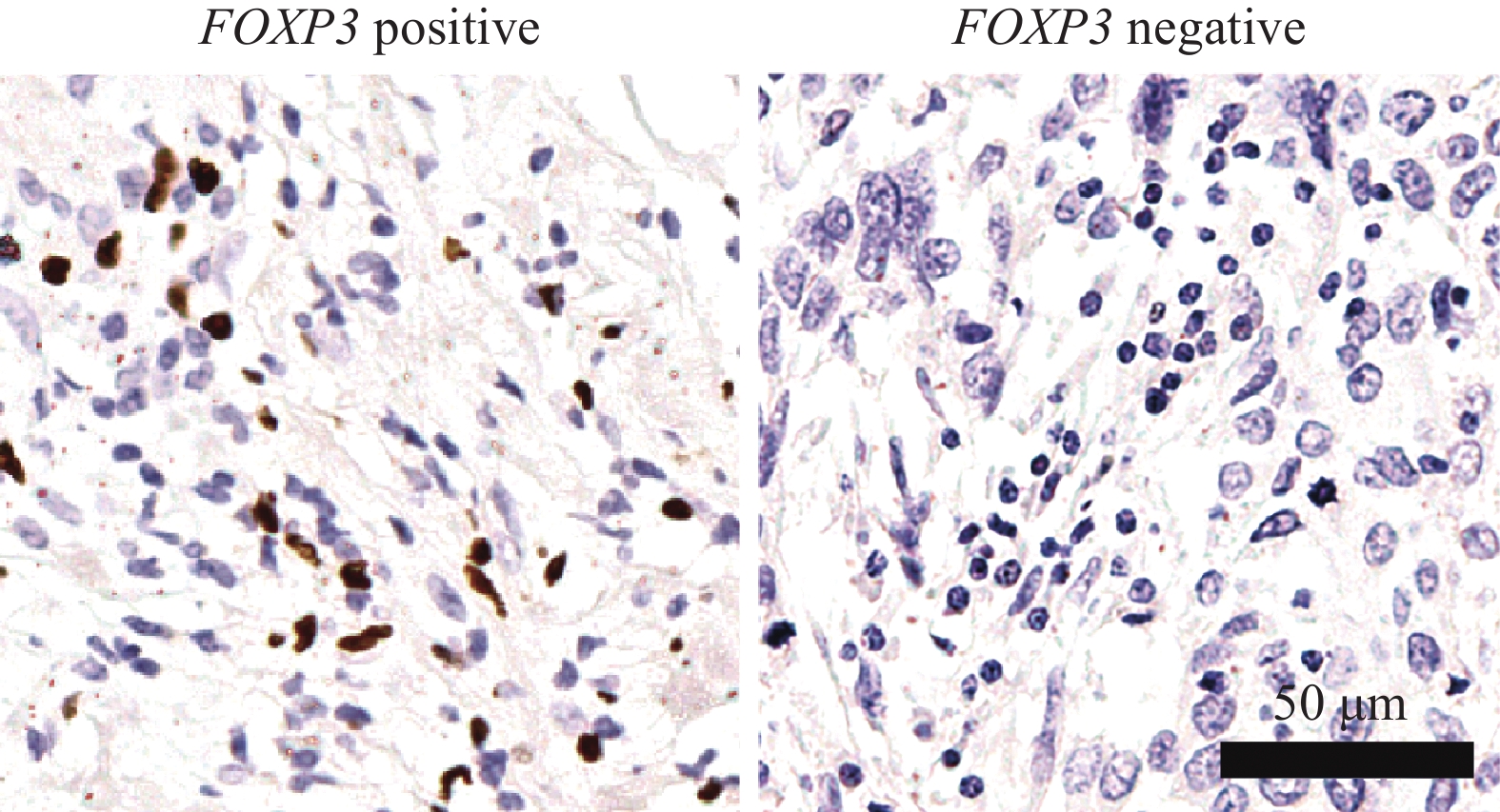
阳性结果判断:FOXP3以乳腺癌浸润淋巴细胞中细胞核出现棕黄色或棕褐色为阳性表达[14-16],而非细胞质浅着色的乳腺癌细胞。每张片子随机选择5个高倍视野(×400),按FOXP3阳性淋巴细胞占所有淋巴细胞的百分比分类,阳性细胞百分比≥5%的表达被定义为FOXP3阳性切片[16]。
1.3 TCGA数据下载和同源重组缺陷评分
癌症基因组图谱乳腺癌组织样本(The Cancer Genome Atlas-Breast Cancer, TCGA-BRCA)的基因组数据从UCSC Xena(http://xena.ucsc.edu/)下载,临床数据从Genomic Data Commons Data Portal(https://gdc-portal.nci.nih.gov/)下载。根据TCGA数据库中乳腺癌患者的全外显子组测序(whole exome sequencing, WES)数据建立同源重组缺陷(homologous recombination deficiency, HRD)评分,计算端粒等位基因失衡(telomeric allelic imbalance, TAI)、大规模状态转变(large-scale state transition, LST)和杂合性缺失(loss of heterozygosity, LOH)评分的总和。
1.4 统计学方法
采用软件R(版本4.2.3)进行数据分析。连续变量的比较采用t检验。分类变量采用Pearson卡方检验或Fisher’s精确检验。相关性分析采用Spearman相关性检验。统计分析采用双侧检验,检验水准α=0.05,P<0.05为差异有统计学意义。
2 结果
2.1 乳腺癌患者的临床病理特征
免疫组织化学法检测纳入的16例BRCA1突变者、32例BRCA2突变者和78例非携带者的乳腺癌组织FOXP3表达,患者年龄、肿瘤分期、淋巴结状态间相互匹配(均P>0.05),见表1。
2.2 FOXP3的表达与BRCA突变状态的关系
FOXP3主要表达于乳腺癌组织T淋巴细胞的细胞核中,见图1。在126例乳腺癌患者中,16例BRCA1突变乳腺癌FOXP3阳性率为43.8%(7/16),32例BRCA2突变乳腺癌FOXP3阳性率为59.4%(19/32),而在78例非携带者乳腺癌仅为9.0%(7/78)。BRCA1突变者和BRCA2突变者乳腺癌FOXP3的阳性率均显著高于非携带者(P=0.002;P<0.001)。进一步在不同分子分型中分析发现,在65例HR+HER2−组中,BRCA1和BRCA2突变携带者FOXP3的阳性率显著高于非携带者(50.0% vs. 5.6%,P=0.04; 64.0% vs. 5.6%,P<0.001)。但在35例TNBC组中,BRCA1和BRCA2突变携带者FOXP3的阳性率与非携带者并未显示出统计学差异(45.5% vs. 20.0%,P=0.22;50.0% vs. 20.0%,P=0.25)。而HER2+组中BRCA1突变携带者(n=1)和BRCA2突变携带者(n=2)的患者数较少,故不作讨论,见表2。
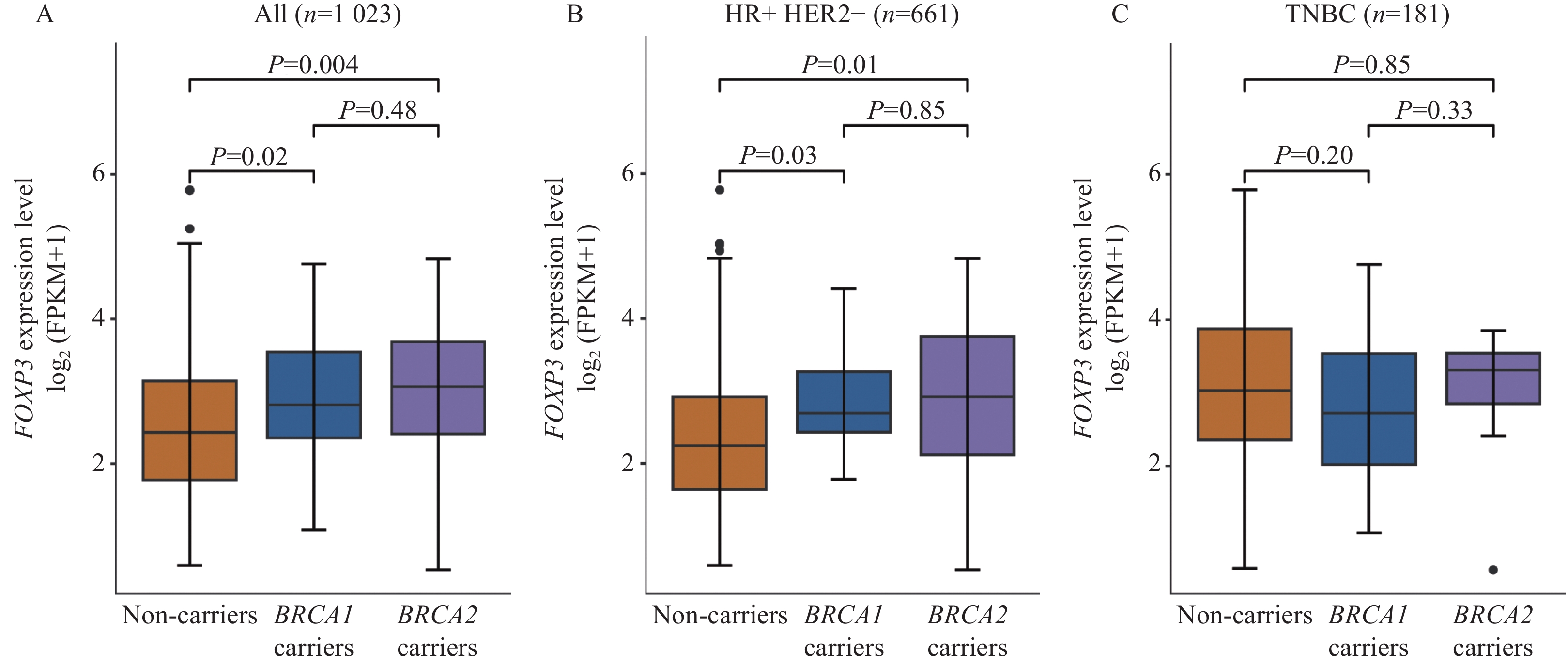
表 2 BRCA1/2突变乳腺癌患者FOXP3表达水平的比较Table 2 Comparison of FOXP3 expression in patients with BRCA1/2-mutant breast cancerMolecular subtype No. FOXP3 level P1 P2 P3 Positive Negative Total 126 33 (26.2%) 93 (73.8%) 0.002 <0.001 0.47 BRCA1 carriers 16 7 (43.8%) 9 (56.3%) BRCA2 carriers 32 19 (59.4%) 13 (40.6%) Non-carriers 78 7 (9.0%) 71 (91.0%) HR+ and HER2− 65 20 (30.8%) 45 (69.2%) 0.04 <0.001 0.62 BRCA1 carriers 4 2 (50.0%) 2 (50.0%) BRCA2 carriers 25 16 (64.0%) 9 (36.0%) Non-carriers 36 2 (5.6%) 34 (94.4%) TNBC 35 11 (31.4%) 24 (68.6%) 0.22 0.25 >0.999 BRCA1 carriers 11 5 (45.5%) 6 (54.5%) BRCA2 carriers 4 2 (50.0%) 2 (50.0%) Non-carriers 20 4 (20.0%) 16 (80.0%) Notes: P1: BRCA1 carriers versus non-carriers; P2: BRCA2 carriers versus non-carriers; P3: BRCA1 carriers versus BRCA2 carriers. 为了验证免疫组织化学的结果,我们比较了TCGA-BRCA数据库中1 023例乳腺癌的RNA表达谱(948例非携带者,39例BRCA1突变携带者,36例BRCA2突变携带者),发现FOXP3的RNA表达水平在BRCA1突变者(P=0.02)和BRCA2突变者(P=0.004)均显著高于非携带者。按照分子分型进行分层分析,在HR+ HER2−组(n=661)中,BRCA1突变者(P=0.03)和BRCA2突变者(P=0.01)的FOXP3表达水平仍显著高于非携带者;而TNBC组(n=181)中未显示出显著差异,这与免疫组织化学的结果一致,见图2。
2.3 FOXP3的表达与同源重组缺陷评分的关系
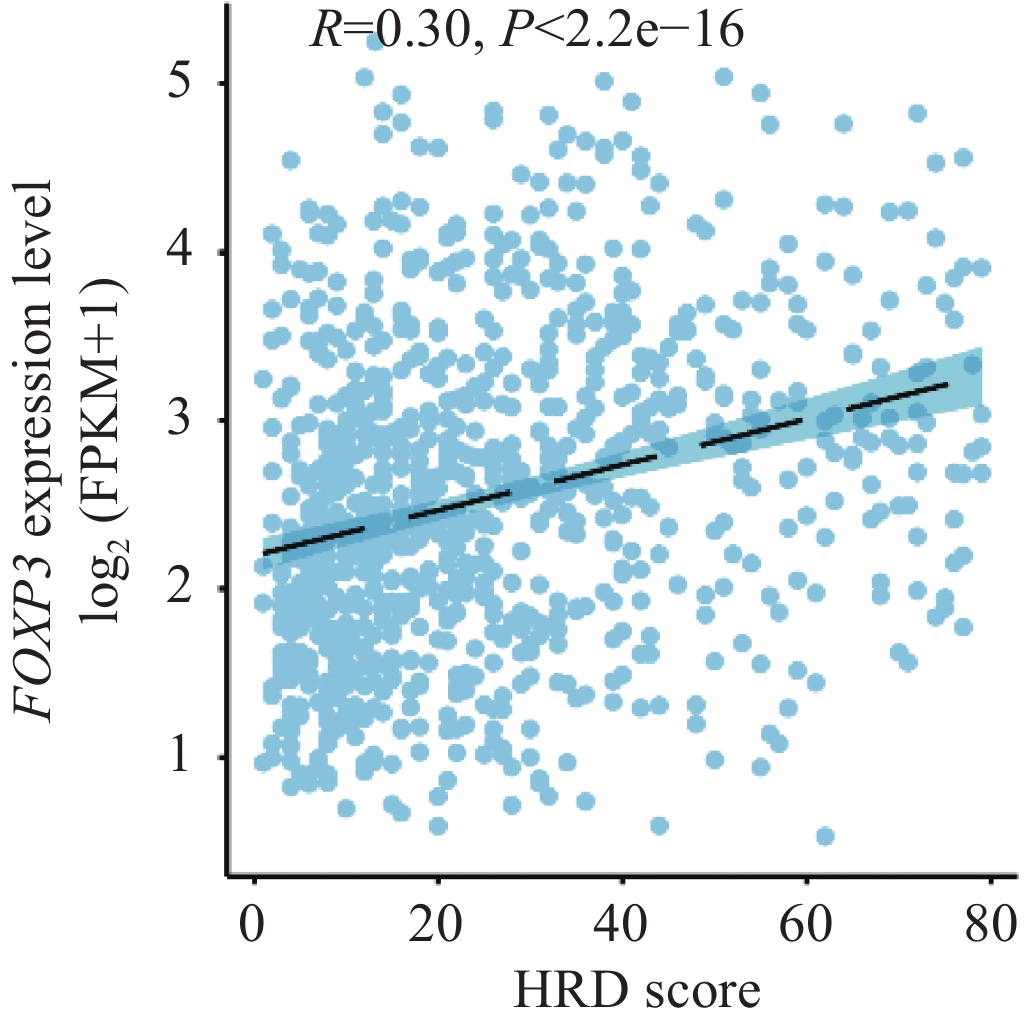
已知BRCA1/2突变携带者同源重组缺陷(HRD)评分比非携带者高[17],在958例具有全外显子测序(WES)的TCGA-BRCA乳腺癌数据中,我们比较了FOXP3的RNA表达水平与HRD评分的关系,发现FOXP3的表达水平与HRD评分正相关(Spearman R=0.30, P<2.2e-16),见图3。
3 讨论
本研究通过免疫组织化学方法发现FOXP3在BRCA1/2突变乳腺癌中表达显著高于非携带者。进一步分析TCGA-BRCA数据发现,FOXP3 RNA表达水平在BRCA1/2突变乳腺癌亦显著高于非突变患者,且FOXP3 RNA表达水平与HRD分数呈正相关。本研究提示Treg细胞在BRCA1/2突变乳腺癌中有较高的富集,并可能与基因组不稳定相关。
FOXP3是一种转录调控因子,对Treg细胞的发育和免疫抑制功能至关重要[11,18]。FOXP3通过结合到特定的DNA序列,调节多个基因的表达,从而影响Treg细胞的分化、稳定性、迁移和免疫抑制能力[11,18-19]。以往关于FOXP3在乳腺癌中的研究并不一致。有文献报道FOXP3阳性Tregs在免疫逃逸中起着重要作用,与乳腺癌生存率低、导管原位癌复发和luminal型乳腺癌中淋巴结转移有关[20-22]。也有研究报道高比例的肿瘤浸润FOXP3阳性细胞与三阴性乳腺癌[23]和BRCA突变乳腺癌[24]的死亡率降低显著相关。以往研究多集中在散发性乳腺癌,目前尚未有研究表明FOXP3表达水平与乳腺癌BRCA突变状态的关系。本研究发现FOXP3阳性淋巴细胞在BRCA突变乳腺癌的富集较非携带者显著增加,表明BRCA突变会刺激更多的Treg细胞富集,BRCA突变乳腺癌较非携带者可能更适合靶向Treg细胞的免疫治疗。进一步按照乳腺癌分子分型分层分析发现,无论在蛋白还是RNA水平,HR+ HER2−的BRCA突变乳腺癌患者FOXP3的表达均高于非携带者,提示HR+ HER2−的BRCA突变患者可能是靶向Treg细胞免疫治疗的潜在受益人群。而在TNBC患者中,BRCA突变者FOXP3阳性率与非携带者之间无统计学差异,可能与该亚组免疫组织化学样本量较小有关,也可能受TNBC本身异质性的免疫微环境影响。
同源重组修复(homologous recombination repair, HRR)是一种高保真的DNA修复途径,以姐妹染色单体作为模板修复DNA双链断裂[25]。同源重组缺陷后DNA双链断裂修复会过度依赖低保真、高易错的替代性DNA损伤修复途径如非同源末端连接,引起大面积染色体重排(如缺失和易位),导致基因组不稳定[25]。BRCA1和BRCA2基因都是参与DNA双链断裂修复的重要因子,它们主要通过HRR方式来维持基因组的稳定性。其中BRCA1在BRCA2的上游发挥作用,BRCA1可以促进DNA端的切割和重组介导因子的结合,为后续的同源配对做准备;BRCA2可以与RAD51结合形成复合体,参与DNA的同源寻找和配对[26]。
HRD状态会导致特定的、可量化的、稳定的基因组改变,包括杂合性缺失、端粒等位不平衡和大片段移位。本研究发现FOXP3的表达水平与基因组不稳定性的指标HRD评分正相关。基因组不稳定性会导致肿瘤细胞产生更多的新生抗原,激活免疫系统,同时也会导致肿瘤细胞产生更多的免疫耐受,抑制免疫系统的攻击。BRCA1/2突变导致的基因组不稳定性和同源重组缺陷,刺激免疫系统的激活和免疫细胞的浸润[9,27]。而具有免疫抑制功能的Treg细胞在BRCA1/2突变乳腺癌中富集,可能是肿瘤细胞为了逃避免疫系统的攻击而产生的一种自我保护的反馈机制,用于抵消免疫系统的攻击,维持肿瘤的免疫耐受状态[9,28]。这一结果提示,基因组不稳定性和Treg细胞水平可能是BRCA1/2突变乳腺癌的免疫治疗的重要预测因子。针对FOXP3或Treg细胞的干预,可能是一种提高BRCA1/2突变乳腺癌免疫治疗效果的有效策略。有研究表明,免疫治疗如抗CTLA-4抗体可以去除Treg细胞的抑制功能,释放效应细胞的细胞毒性功能[12-13,29]。提示BRCA1/2突变乳腺癌可能受益于靶向Treg细胞的免疫治疗。
总之,本研究发现BRCA突变乳腺癌中FOXP3表达阳性率高,提示BRCA突变乳腺癌FOXP3高表达者可能是免疫治疗潜在的获益人群。未来还需要更大样本验证本结果,以及体内外功能实验来揭示FOXP3在BRCA1/2突变乳腺癌中的作用机制,为BRCA1/2突变乳腺癌的免疫治疗提供理论依据。
Competing interests: The authors declare that they have no competing interests.利益冲突声明:所有作者均声明不存在利益冲突。作者贡献:陈琳茜:实施实验,分析数据,撰写论文胡丽、陈久安、姚璐、张娟:收集整理数据徐 晔:收集整理数据,修改论文解云涛:指导研究方案,修改论文 -
表 1 BRCA1/2突变乳腺癌患者和非携带者临床病理特征
Table 1 Clinicopathological characteristics of patients with BRCA1/2-mutant breast cancer and non-carriers
Characteristics No. BRCA1 carriers (n(%)) BRCA2 carriers (n(%)) Non-carriers (n(%)) P1 P2 P3 Total 126 16 32 78 Median follow-up
time (range, years)4.7 (0.1-19.2) 8.2 (1.5-19.2) 9.2 (0.1-18.2) 4.0 (0.4-5.1) Age at diagnosis (years) Median (range) 49 (27-75) 43.5 (28-64) 49.5 (31-74) 50 (27-75) ≤ 50 68 12 (75.0%) 16 (50.0%) 40 (51.3%) 0.14 >0.999 0.18 > 50 58 4 (25.0%) 16 (50.0%) 38 (48.7%) ER status Positive 74 5 (31.3%) 24 (75.0%) 45 (57.7%) 0.10 0.14 0.009 Negative 52 11 (68.8%) 8 (25.0%) 33 (42.3%) PR status Positive 65 5 (31.3%) 26 (81.3%) 34 (43.6%) 0.52 < 0.001 0.002 Negative 61 11 (68.8%) 6 (18.8%) 44 (56.4%) HER2 status Positive 26 1 (6.3%) 3 (9.4%) 22 (28.2%) 0.11 0.06 >0.999 Negative 100 15 (93.8%) 29 (90.6%) 56 (71.8%) Molecular type TNBC 35 11 (68.8%) 4 (12.5%) 20 (25.6%) 0.01 0.008 < 0.001 HR+ and HER2− 65 4 (25.0%) 25 (78.1%) 36 (46.2%) HER2+ 26 1 (6.3%) 3 (9.4%) 22 (28.2%) Tumor stage Ⅰ 29 5 (31.3%) 9 (28.1%) 15 (19.2%) 0.21 0.23 0.67 Ⅱ 77 9 (56.3%) 14 (43.8%) 54 (69.2%) Ⅲ 12 0 (0) 3 (9.4%) 9 (11.5%) Unknown 8 2 (12.5%) 6 (18.8%) 0 (0) Tumor size (cm) ≤2 50 6 (37.5%) 19 (59.4%) 25 (32.1%) 0.56 0.01 0.35 >2 75 9 (56.3%) 13 (40.6%) 53 (67.9%) Unknown 1 1 (6.3%) 0 (0) 0 (0) Nodal status Positive 41 4 (25.0%) 12 (37.5%) 25 (32.1%) 0.77 0.79 0.59 Negative 85 12 (75.0%) 20 (62.5%) 53 (67.9%) Notes: P1: BRCA1 carriers versus non-carriers; P2: BRCA2 carriers versus non-carriers; P3: BRCA1 carriers versus BRCA2 carriers. ER: estrogen receptor; PR: progesterone receptor; HER2: human epidermal growth factor receptor 2; TNBC: triple-negative breast cancer; HR: hormone receptor. 表 2 BRCA1/2突变乳腺癌患者FOXP3表达水平的比较
Table 2 Comparison of FOXP3 expression in patients with BRCA1/2-mutant breast cancer
Molecular subtype No. FOXP3 level P1 P2 P3 Positive Negative Total 126 33 (26.2%) 93 (73.8%) 0.002 <0.001 0.47 BRCA1 carriers 16 7 (43.8%) 9 (56.3%) BRCA2 carriers 32 19 (59.4%) 13 (40.6%) Non-carriers 78 7 (9.0%) 71 (91.0%) HR+ and HER2− 65 20 (30.8%) 45 (69.2%) 0.04 <0.001 0.62 BRCA1 carriers 4 2 (50.0%) 2 (50.0%) BRCA2 carriers 25 16 (64.0%) 9 (36.0%) Non-carriers 36 2 (5.6%) 34 (94.4%) TNBC 35 11 (31.4%) 24 (68.6%) 0.22 0.25 >0.999 BRCA1 carriers 11 5 (45.5%) 6 (54.5%) BRCA2 carriers 4 2 (50.0%) 2 (50.0%) Non-carriers 20 4 (20.0%) 16 (80.0%) Notes: P1: BRCA1 carriers versus non-carriers; P2: BRCA2 carriers versus non-carriers; P3: BRCA1 carriers versus BRCA2 carriers. -
[1] Sung H, Ferlay J, Siegel RL, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries[J]. CA Cancer J Clin, 2021, 71(3): 209-249. doi: 10.3322/caac.21660
[2] Zang F, Ding X, Chen J, et al. Prevalence of BRCA1 and BRCA2 pathogenic variants in 8627 unselected patients with breast cancer: stratification of age at diagnosis, family history and molecular subtype[J]. Breast Cancer Res Treat, 2022, 195(3): 431-439. doi: 10.1007/s10549-022-06702-4
[3] Mahdavi M, Nassiri M, Kooshyar MM, et al. Hereditary breast cancer; Genetic penetrance and current status with BRCA[J]. J Cell Physiol, 2019, 234(5): 5741-5750. doi: 10.1002/jcp.27464
[4] Kuchenbaecker KB, Hopper JL, Barnes DR, et al. Risks of Breast, Ovarian, and Contralateral Breast Cancer for BRCA1 and BRCA2 Mutation Carriers[J]. JAMA, 2017, 317(23): 2402-2416. doi: 10.1001/jama.2017.7112
[5] Liu Y, Hu Y, Xue J, et al. Advances in immunotherapy for triple-negative breast cancer[J]. Mol Cancer, 2023, 22(1): 145. doi: 10.1186/s12943-023-01850-7
[6] Keenan TE, Tolaney SM. Role of Immunotherapy in Triple-Negative Breast Cancer[J]. J Natl Compr Canc Netw, 2020, 18(4): 479-489. doi: 10.6004/jnccn.2020.7554
[7] de Boo L, Cimino-Mathews A, Lubeck Y, et al. Tumour-infiltrating lymphocytes (TILs) and BRCA-like status in stage Ⅲ breast cancer patients randomised to adjuvant intensified platinum-based chemotherapy versus conventional chemotherapy[J]. Eur J Cancer, 2020, 127: 240-250. doi: 10.1016/j.ejca.2019.12.003
[8] Wen WX, Leong CO. Association of BRCA1- and BRCA2-deficiency with mutation burden, expression of PD-L1/PD-1, immune infiltrates, and T cell-inflamed signature in breast cancer[J]. PLoS One, 2019, 14(4): e0215381. doi: 10.1371/journal.pone.0215381
[9] van Vugt MATM, Parkes EE. When breaks get hot: inflammatory signaling in BRCA1/2-mutant cancers[J]. Trends Cancer, 2022, 8(3): 174-189. doi: 10.1016/j.trecan.2021.12.003
[10] Qiu R, Zhou L, Ma Y, et al. Regulatory T Cell Plasticity and Stability and Autoimmune Diseases[J]. Clin Rev Allergy Immunol, 2020, 58(1): 52-70. doi: 10.1007/s12016-018-8721-0
[11] Ramsdell F, Rudensky AY. Foxp3: a genetic foundation for regulatory T cell differentiation and function[J]. Nat Immunol, 2020, 21(7): 708-709. doi: 10.1038/s41590-020-0694-5
[12] Tanaka A, Sakaguchi S. Regulatory T cells in cancer immunotherapy[J]. Cell Res, 2017, 27(1): 109-118. doi: 10.1038/cr.2016.151
[13] Li C, Jiang P, Wei S, et al. Regulatory T cells in tumor microenvironment: new mechanisms, potential therapeutic strategies and future prospects[J]. Mol Cancer, 2020, 19(1): 116. doi: 10.1186/s12943-020-01234-1
[14] Ilie SM, Briot N, Constatin G, et al. Pathologic and immunohistochemical prognostic markers in residual triple-negative breast cancer after neoadjuvant chemotherapy[J]. Front Oncol, 2024, 13: 1309890. doi: 10.3389/fonc.2023.1309890
[15] Glajcar A, Łazarczyk A, Tyrak KE, et al. Nodal status in luminal A invasive breast cancer: relationships with cytotoxic CD8 + and regulatory FOXP3 + cells tumor-associated infiltrate and other prognostic factors[J]. Virchows Arch, 2021, 479(5): 871-882. doi: 10.1007/s00428-021-03126-1
[16] Kalaw E, Lim M, Kutasovic JR, et al. Metaplastic breast cancers frequently express immune checkpoint markers FOXP3 and PD-L1[J]. Br J Cancer, 2020, 123(11): 1665-1672. doi: 10.1038/s41416-020-01065-3
[17] Timms KM, Abkevich V, Hughes E, et al. Association of BRCA1/2 defects with genomic scores predictive of DNA damage repair deficiency among breast cancer subtypes[J]. Breast Cancer Res, 2014, 16(6): 475. doi: 10.1186/s13058-014-0475-x
[18] Bin Dhuban K, d'hennezel E, Nagai Y, et al. Suppression by human FOXP3(+) regulatory T cells requires FOXP3-TIP60 interactions[J]. Sci Immunol, 2017, 2(12): eaai9297. doi: 10.1126/sciimmunol.aai9297
[19] Kwon HK, Chen HM, Mathis D, et al. Different molecular complexes that mediate transcriptional induction and repression by FoxP3[J]. Nat Immunol, 2017, 18(11): 1238-1248. doi: 10.1038/ni.3835
[20] Núñez NG, Tosello Boari J, Ramos RN, et al. Tumor invasion in draining lymph nodes is associated with Treg accumulation in breast cancer patients[J]. Nat Commun, 2020, 11(1): 3272. doi: 10.1038/s41467-020-17046-2
[21] Merlo A, Casalini P, Carcangiu ML, et al. FOXP3 expression and overall survival in breast cancer[J]. J Clin Oncol, 2009, 27(11): 1746-1752. doi: 10.1200/JCO.2008.17.9036
[22] Semeraro M, Adam J, Stoll G, et al. The ratio of CD8(+)/FOXP3 T lymphocytes infiltrating breast tissues predicts the relapse of ductal carcinoma in situ[J]. Oncoimmunology, 2016, 5(10): e1218106. doi: 10.1080/2162402X.2016.1218106
[23] West NR, Kost SE, Martin SD, et al. Tumour-infiltrating FOXP3(+) lymphocytes are associated with cytotoxic immune responses and good clinical outcome in oestrogen receptor-negative breast cancer[J]. Br J Cancer, 2013, 108(1): 155-162. doi: 10.1038/bjc.2012.524
[24] Jørgensen N, Hviid TVF, Nielsen LB, et al. Tumour-infiltrating CD4−, CD8− and FOXP3-positive immune cells as predictive markers of mortality in BRCA1- and BRCA2-associated breast cancer[J]. Br J Cancer, 2021, 125(10): 1388-1398. doi: 10.1038/s41416-021-01514-7
[25] Vollebergh MA, Jonkers J, Linn SC. Genomic instability in breast and ovarian cancers: translation into clinical predictive biomarkers[J]. Cell Mol Life Sci, 2012, 69(2): 223-245. doi: 10.1007/s00018-011-0809-0
[26] Roy R, Chun J, Powell SN. BRCA1 and BRCA2: different roles in a common pathway of genome protection[J]. Nat Rev Cancer, 2011, 12(1): 68-78.
[27] Jiang M, Jia K, Wang L, et al. Alterations of DNA damage response pathway: Biomarker and therapeutic strategy for cancer immunotherapy[J]. Acta Pharm Sin B, 2021, 11(10): 2983-2994. doi: 10.1016/j.apsb.2021.01.003
[28] Wing JB, Tanaka A, Sakaguchi S. Human FOXP3(+) Regulatory T Cell Heterogeneity and Function in Autoimmunity and Cancer[J]. Immunity, 2019, 50(2): 302-316. doi: 10.1016/j.immuni.2019.01.020
[29] Tay C, Tanaka A, Sakaguchi S. Tumor-infiltrating regulatory T cells as targets of cancer immunotherapy[J]. Cancer Cell, 2023, 41(3): 450-465. doi: 10.1016/j.ccell.2023.02.014
-
期刊类型引用(1)
1. 蔡钊萌,邝枣园. 宝藿苷Ⅰ抑制调节性T细胞FoxP3表达及其作用机制. 中国临床药理学杂志. 2024(24): 3590-3594 .  百度学术
百度学术
其他类型引用(0)



 下载:
下载: